Disinfectant Residues: Mitigation and Management
Problems with Residues
Cleanroom residues can be generated by the process, raw materials, and people, but a significant contributor to the residues in a cleanroom are the disinfectants themselves. Disinfectants residue is anything left behind by the disinfectant on surfaces, equipment, windows, etc. Residues are inevitable when using a disinfectant.
Cleaning and disinfection are often two words that are used interchangeably, when in fact, they have two different meanings. Cleaning is physically removing organic matter from surface. Disinfection is the use of antimicrobial agents to kill microorganisms on the surface. Residue removal is specific to cleaning.
Most commonly used disinfectants leave significant residues on a surface, which can subsequently have a detrimental effect microbial control in the cleanroom. This is acknowledged in United States Pharmacopeia (USP) <1072>, the Parenteral Drug Association’s Technical Report 70 (PDA TR 70), and Eudralex Volume 4 Good Manufacturing Guidelines (GMP) Annex I.
In cleanrooms, disinfectant residues are often monitored, or measured, visually. Disinfectant residues can cause visual, safety, and product integrity threats, including sticky or slippery floors and doors, streaks and discoloration, and contamination. If residues are not managed correctly, they can also cause degradation to the facility over time, which can lead to costly reconstruction or require deep cleaning measures.
Disinfectants are Crucial in Residue Management
Application of disinfectants and proper use of tools such as wipers and mops play a critical role in residue management and there are many variables that can pose issues when cleaning and disinfecting including over-application of disinfectants, frequency of residue removal, reapplication of disinfectant (if longer contact times are required), and the nature of the disinfectant. There is no one-size-fits-all approach for mitigation of residues as part of an overall Contamination Control Strategy (CCS).
The appearance of disinfectant residue on different surfaces within the cleanroom can also look lesser or greater for the same amount of residue dependent on the characteristics of the surface itself. It is common to see disinfectant residues on windows, which can make a facility look dirty and unkept. Highly reflective surfaces, such as glass and polished metals, will show the residue more significantly, although the amount of residue will be the same.
A disinfectant residue removal step is necessary to mitigate the residue and maintain cleanliness of the surfaces within the cleanroom. It can include the use of 70% Isopropyl Alcohol (IPA) solution or presaturated wipe, Water for Injection (WFI) or Purified Water (PW), 6% Hydrogen Peroxide (6% H202), a detergent, or even simply a dry wipe. If using a detergent, is it is recommended to rinse with water and follow with a dry wipe (or mop) to remove the solubilized residue since detergents, like disinfectants, contain surfactants that will also leave residues.
Periodically Rinse to Prevent Accumulation
While such a rinse step is likely not to be required after every cleaning event, one must be included periodically to prevent recurrence of residue accumulation on all cleaned surfaces. A reasonable period for residue removal could be every two weeks, or once per month depending on the specific disinfectant chemistries used, the frequency of application, and amount of disinfectant applied each cleaning. This is also dependent on a risk assessment to the specific cleanroom. It is always recommended to start with longer periods of time between cleanings and adjust as needed. Additionally, a detergent may be needed when disinfectant residue is hard to remove, or has accumulated on the surface for a long period of time.
Residue on Evaporation (RoE)
A common method for measuring the potential for residues is called “Residue on Evaporation” (RoE). It involves weighing a solution (100 mL) of the disinfectant, then slowly heating the disinfectant to 104°C for 4-18 hours. After cooling, the sample is weighed again. The difference in weight is expressed as ppm (mg/liter). This method was conducted on several commercial disinfectants by a third-party laboratory (ALS). Below are the results from this study.
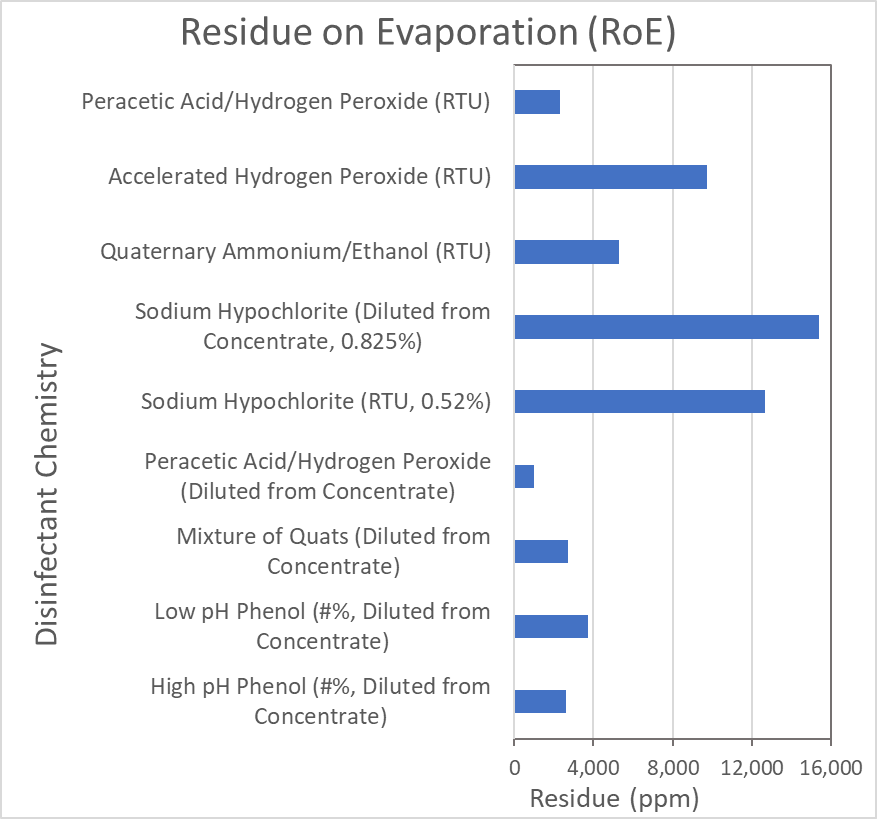
As can be seen from the results, the level of residue for the different active ingredients can vary significantly from product to product. The table also shows there can be a difference for disinfectants containing the same active ingredient, which would be due to the concentration of the active ingredient in the product, the amount of stabilizers required, and potentially the addition of preservatives, pH adjusters or odor mitigators.
Visual appearance of a disinfectant residue on a surface may not always correlate with the amount of residue indicated by a residue on evaporation test. Since all disinfectant residues are not the same, it is also important to understand how readily the disinfectant residue is removed from equipment, surfaces, floors, walls, ceilings and windows.
To determine the best course of removal, these simple steps can be taken:
- Evaluate the severity of the residue
- How long it has been on the surface
- Look and feel of surfaces
- Disinfectant characteristics
- Determine what removal aide to be used for disinfectant removal:
- Detergent
- Water for Injection (WFI)
- 70% Isopropyl Alcohol (IPA)
- 6% Hydrogen Peroxide
- Dry wipe
- Implement removal aide as part of cleaning and disinfection program
- Determine frequency of removal aide as part of the cleaning and disinfection program
General Guidelines for Removal of Disinfectant Residues
There are many options for developing a residue removal step to a cleaning and disinfection program. Below are examples of steps that can be taken to remove residues from the controlled area or cleanroom. These are general recommendations and are to be used as guidance independent of the disinfectant used. Always follow site Standard Operating Procedures (SOPs).
Solutions used may be dependent upon the surfaces to be cleaned, environmental versus product contact.
Residue Removal Steps with Dry Wipe on Wet Surface:
- Allow disinfectant to meet validated wet contact time.
- Wipe wet disinfectant with dry wipe or mopping tool.
- Remove excess residue with 70% presaturated wipe.
Residue Removal Steps with Presaturated Wipe on Dry Surfaces:
- Allow disinfectant to meet validated wet contact time.
- Let surface dry.
- Remove disinfectant residue with presaturated 70% IPA wipe (may have to be repeated depending on the severity of the residue).
Residue Removal Step for Floors, Walls, Ceilings for Dry Surfaces with WFI:
- Allow disinfectant to meet validated wet contact time.
- Let surface dry.
- Apply WFI to surface using mop or sprayer (if sprayer is used, ensure water does not get into hard to reach places, if there is a risk for that, it is not recommended).
- Mop or vacuum until dry.
Residue Removal Step for Floors, Walls, Ceilings for Dry Surfaces with Detergent:
- Allow disinfectant to meet validated wet contact time.
- Let surface dry.
- Apply detergent to surface using mop.
- WFI rinse with mop.
- Follow up dry mop or vacuum.
Residue Removal Step for Floors, Walls, Ceilings on Wet Surfaces:
- Allow disinfectant to meet validated wet contact time.
- Follow with dry mop or vacuum
Disinfectant residues can pose significant cleanliness and operational risks in cleanrooms and other controlled environments, in additional to degrading the appearance of cleanliness. Regulatory organizations worldwide recognize the need to remove or otherwise mitigate disinfectant residues as part of an effective overall Contamination Control Strategy.
It is critical to understand the nature of disinfectant residues in order to establish an effective residue management strategy and program. Creation and implementation of effective, simple protocols for residue management result in a cleanroom that is cleaner, safer, and easier to maintain in microbial and contamination control.
References:
